Reactivity periodic table4/19/2023 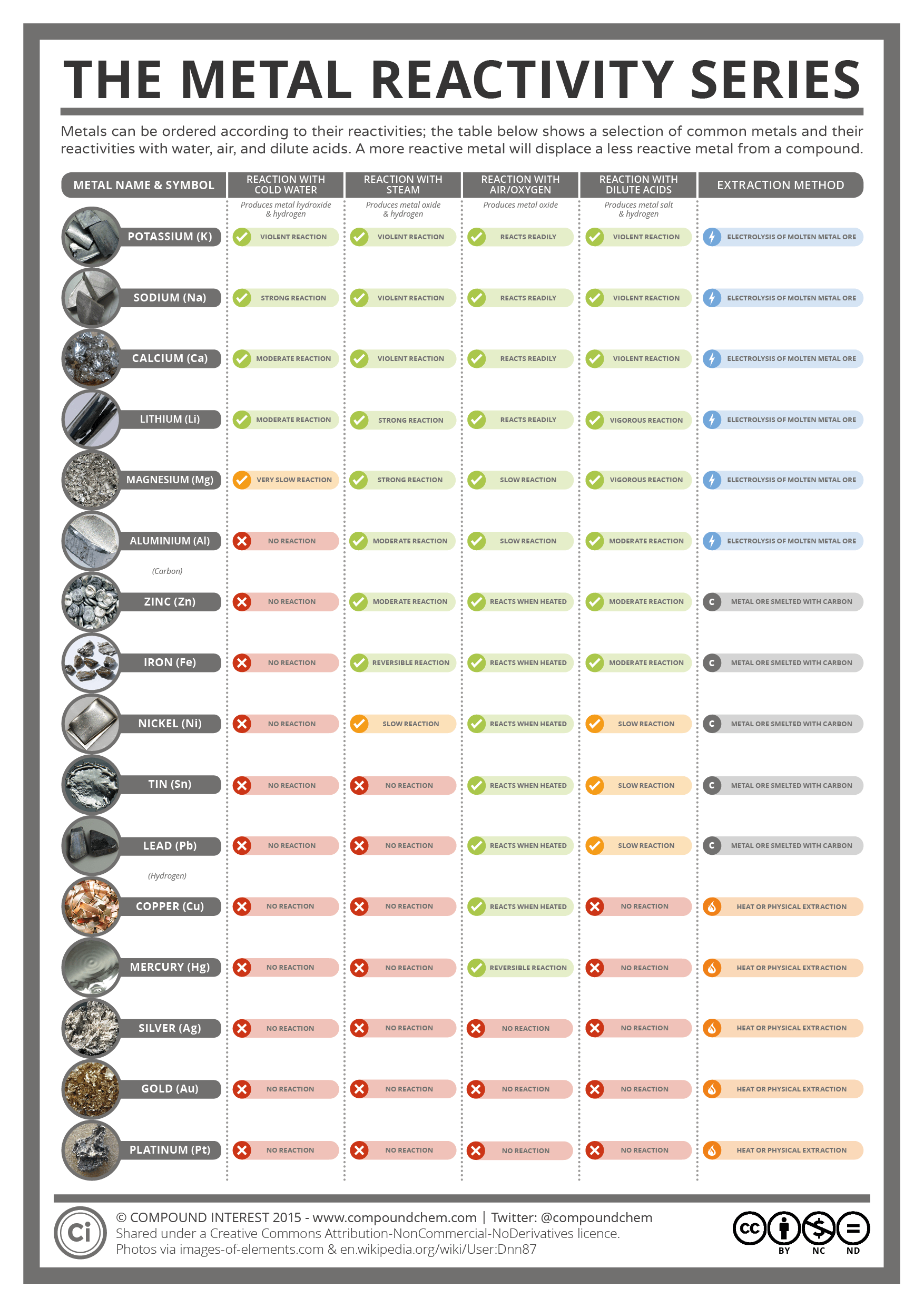
Further information is provided in our H ealth and Safety guidance. Do NOT follow suggestions which conflict with current advice from CLEAPSS , SSERC or recent safety guides. eLibrary users are responsible for ensuring that any activity, including practical work, which they carry out is consistent with current regulations related to h ealth and s afety and that they carry an appropriate risk assessment. Visit the secondary science webpage to access all lists: Whilst this list provides a source of information and ideas for experimental work, it is important to note that recommendations can date very quickly. This video explains trends in the atomic radii of elements in the periodic. The periodic table has two groups metal and non-metal elements and so to answer your question I will focus on each group and identify the most reactive element. 
the order of metals and carbon in the reactivity series state the trend in chemical reactivity : I) across the period left to right.how patterns in reactions can be predicted with reference to the periodic table.The resources link to the following topics : This list provides some advice on carrying out some traditional demonstrations and ideas for practicals which which explore reactivity. Thus, if we may see the Li-family.1 answer Top answer: Im assuming youre asking about how the CHARACTER changes as we move all about the periodic table, and based on that how does the reactivity change. 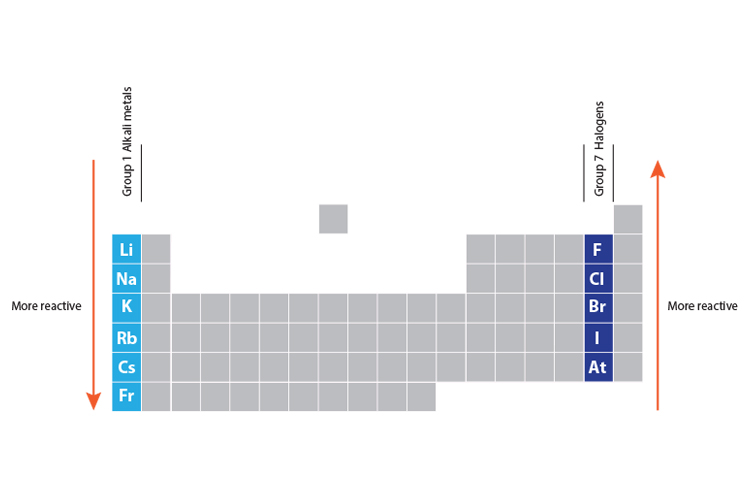
Practical activities which look at the reactions of different groups of elements are always exciting. When we go down a group, electropositive character increases, but electronegative character decreases. Patterns in reactions, with reference to the periodic table
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
